(Radium-224 is a decay product of thorium, and it decays to radon-220, also known as thoron, with a 54-s half-life.)Because radon is so short-lived, and alpha-decays to a number of daughter products, which are solid and very short-lived, there is a high probability of its decay when breathed in, or when radon daughter products in dust are breathed in. When this radium decays it produces radon-222, an inert gas with a half-life of almost 4 days. Radium-226 is one of the decay products of uranium-238, which is widespread in most rocks and soils. Of course, physical measurements to which tracer results are compared to test the success of an isotopic method are themselves subject to a certain amount of inaccuracy, and should be evaluated for accuracy similar to the tracer data.īahman Zohuri, in Nuclear Reactor Technology Development and Utilization, 2020 2.9.14 Radon Regardless of which case is closer to the truth, the mixing proportions are in good agreement with those calculated from gauge measurements under either condition discussed above. Thus, depending on the value of Qused as Cleghorn Springs water inflow into Rapid Creek, the result is either 7.9 cfs or 10.4 cfs. If, on the other hand, the water was lost in the more considerable distance between the upstream gage and Cleghorn Springs, the downstream water represents 66% upstream water and 34% Cleghorn Springs water. If it is assumed that the imbalance is lost through the stream bed after complete mixing with the Cleghorn Springs water, the downstream end-member represents 69% upstream water and 31 % Cleghorn Springs water. Or the total flow ( Q d) was about 31 % Cleghorn Springs water, and 69% upstream Rapid Creek water.įrom Table 20.7 it can be seen that the gauge values of the flow measuring stations are not internally consistent in that the sum of the two inputs is 3.2 cubic feet per second larger than the output. The lower volume and higher contaminant concentration in the retentate would improve the efficiency of the precipitation process, compared to treating the original groundwater however, these savings would need to offset the cost of the RO treatment. Precipitation could be used to treat the retentate. 
RO will remove Ra from water, along with all of the other ions. The addition of sulfate ions along with the Ba can further improve Ra precipitation, to even below the true solubility of RaSO 4, apparently due to adsorption of the Ra on the surface of the BaSO 4 particles ( Doerner & Hoskins, 1925). The addition of barium ions prior to increasing the pH will further reduce the Ra concentration, due to co-precipitation with the barium. Radium is insoluble at high pH, so precipitation can be used to treat groundwater. The concentration of natural Sr, and Ba if present, would be much higher than the Ra concentration and would determine the amount of water that could be treated prior to Ra breakthrough. Zeolites have an affinity for Ra, similar to strontium and barium ions ( Chalupnik, Franus, & Gzyl, 2013). Removal of over 90% of the Ra from groundwater can be achieved by cation exchange ( Brinck, Schliedelman, Bennett, Bell, & Markwood, 1976). 
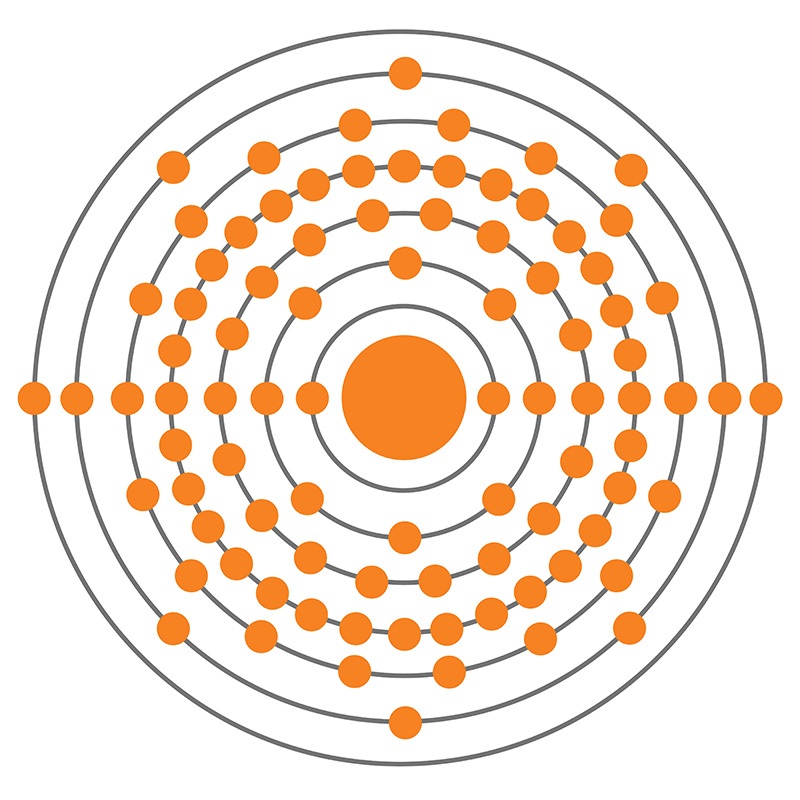
Radium is likely to break through an ion exchange column only slightly after Ca and Mg, so the amount of groundwater that can be treated before regeneration or disposal of the resin is required will mostly be driven by the concentration of Ca and Mg in the water. Strong acid–cation exchange resins have a much higher affinity for Ra over Na and a slightly higher affinity for Ra over Mg and Ca. Some of the 226Ra is recovered for commercial use. Radium-226 is a byproduct of uranium mining, either left in ore that is leached with carbonate to recover the uranium or leached from the ore along with the uranium if sulfuric acid is used. The chemistry of radium is similar to calcium, magnesium, and strontium. The natural concentration of Ra in groundwater can vary widely, depending on the local concentration of uranium and thorium. The half-life of the radium isotopes are 1599 years for 226Ra and 5.76 years for 228Ra. Groundwater in the vicinity of these secondary deposits would contain high concentrations of 226Ra ( Cothem & Rebers, 1990). If reducing conditions are encountered, the uranium can precipitate, forming concentrated secondary deposits. Thorium is more abundant than uranium, so 228Ra is generally present in higher concentrations however, uranium complexes are soluble in water in oxidizing conditions and can move with the groundwater. Radium occurs naturally in very low concentrations from the decay of uranium and thorium, 226Ra from 238U and 228Ra from 232Th. Taylor, in Environmental Remediation and Restoration of Contaminated Nuclear and Norm Sites, 2015 10.5.3 Radium 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
